
Quality & Compliance Documentation
Keep quality records audit-ready across plants and partners
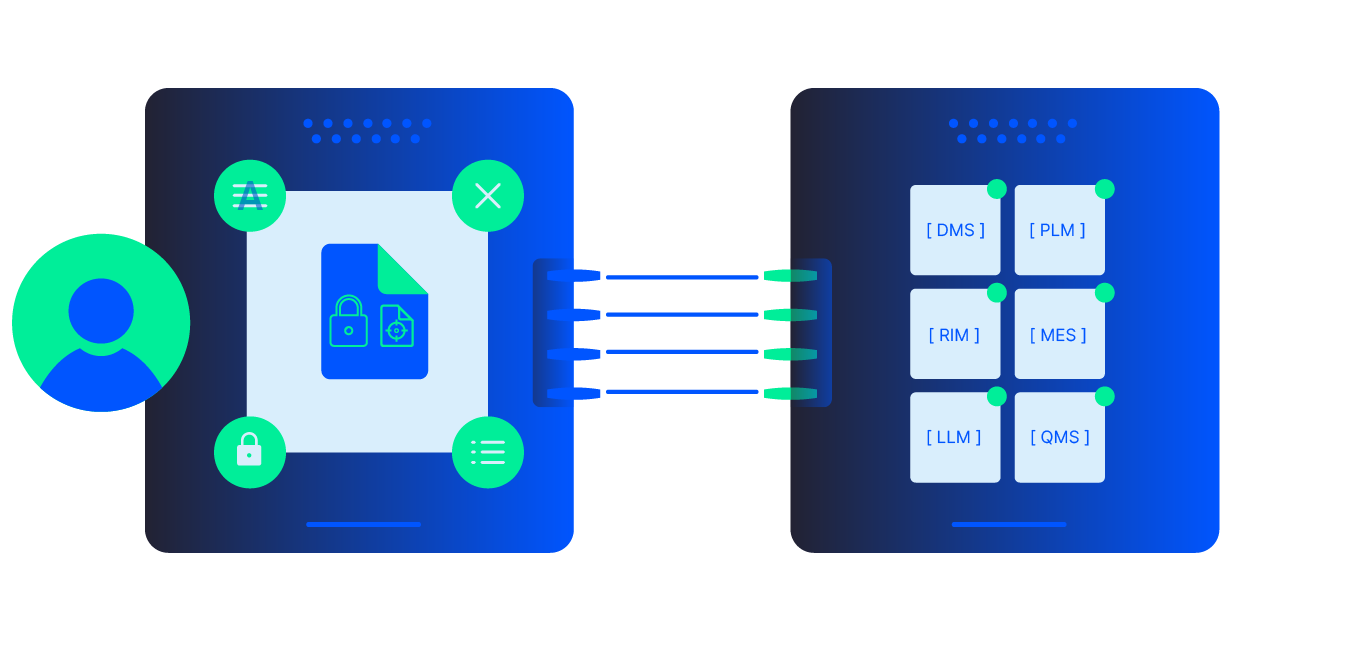
Standardize and automate the documents that underpin quality and regulatory compliance - SOPs, work instructions, batch records, deviations, CAPAs, and inspection reports. Adlib ingests, normalizes, and assembles these into consistent, searchable documents of record, making it easier to pass audits, reduce deviations, and prove compliance across every site, line, and supplier.








.png)
.png)
