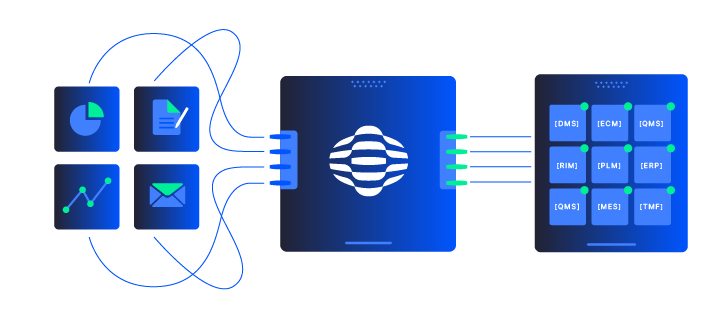
Your use case is usually a strong fit for Adlib if it checks three boxes:
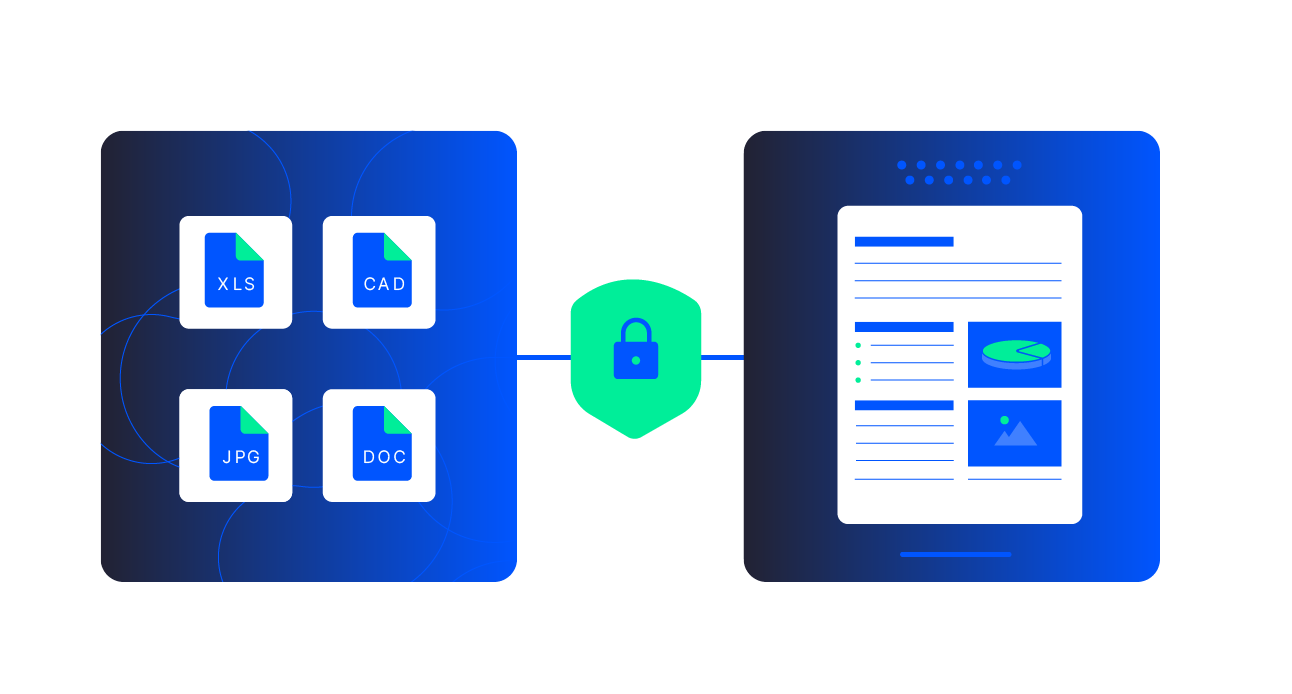
- Document-heavy and multi-format (scans, PDFs, emails, legacy files, CAD),
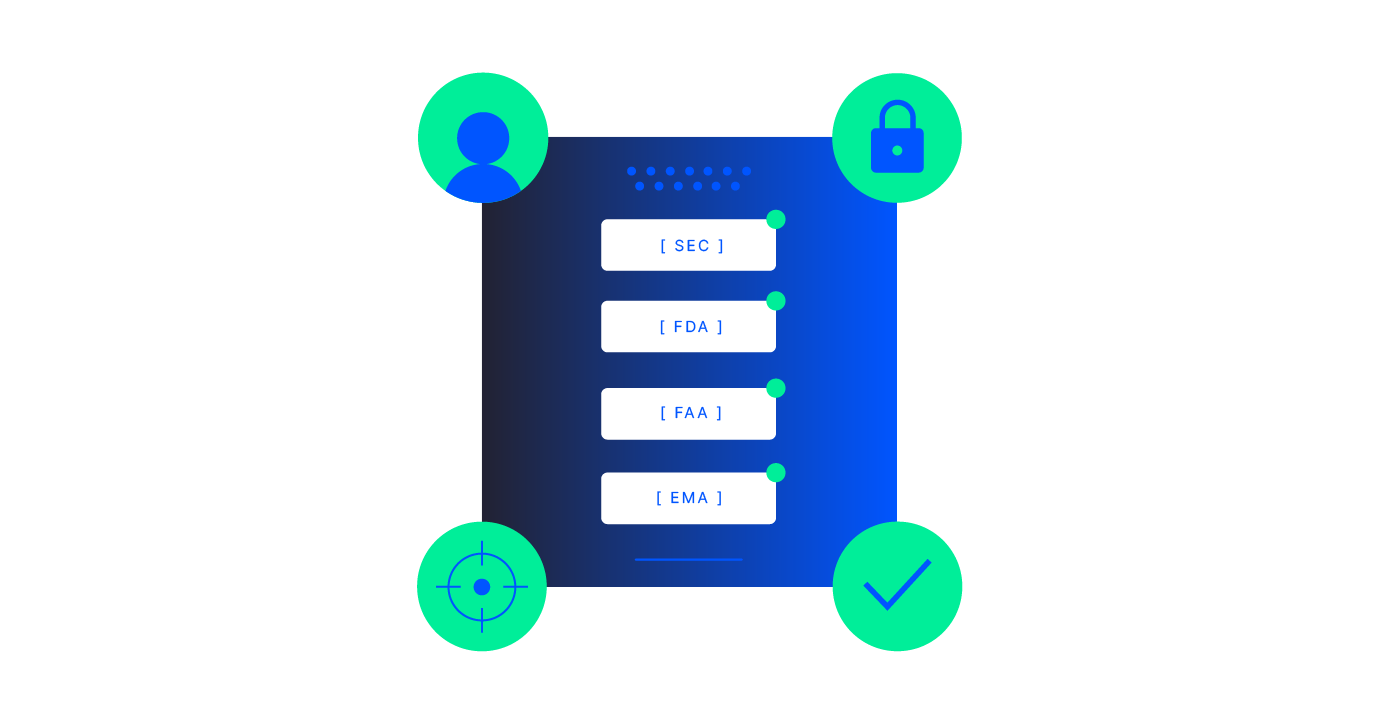
- High-stakes for accuracy and compliance (regulators, auditors, safety, financial risk), and
- Currently dependent on manual review or stitching multiple tools together.
If you have workflows like regulatory submissions, inspections, claims, incident reports, FOI/ATI responses, quality and batch records, or KYC and loan packages, Adlib can help you build an accuracy-first, AI-ready pipeline around them. Many organizations start with a single painful workflow, then expand to adjacent use cases once results are proven.





















.png)
.png)
.png)